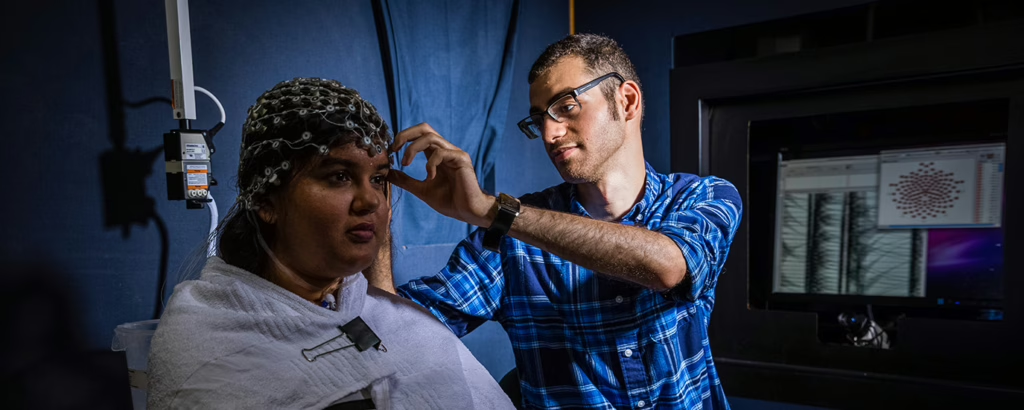
Clark University’s Institutional Review Board (IRB) is responsible for ensuring that all research conducted by faculty, staff, and students, protects the rights and welfare of human subjects. All human subjects research at Clark must be reviewed by the IRB.

CITI Training
All individuals submitting proposals to the IRB must have completed CITI training. Training must be renewed every three years. Mentor will automatically flag your submission if your training has expired. CITI training must include the “RCR Basic Curriculum + Human Subjects” modules.



